ABC Chemistry – Best Chemistry Coaching in Dwarka, Delhi

Master Organic Chemistry with Mechanisms & Reaction Strategy
Comprehensive coverage of reaction mechanisms, retrosynthesis, and synthetic strategies by ABC Chemistry for CSIR-NET, IIT-JAM, GATE & CUET-PG.
Why Organic Chemistry at ABC Chemistry?
Organic Chemistry becomes easy when reactions are understood through electron flow, intermediates, and mechanisms. Our teaching focuses on concept clarity instead of memorization, helping students solve even unfamiliar questions confidently.
Organic Chemistry Syllabus
GOC
Inductive, resonance, hyperconjugation, acidity & basicity.
Stereochemistry
R/S, E/Z, optical activity, conformations.
Reaction Mechanism
SN1, SN2, E1, E2, rearrangements.
Aromatic Chemistry
Electrophilic substitution & directing effects.
Carbonyl Chemistry
Aldehydes, ketones, acids & derivatives.
Named Reactions
Pericyclic & advanced transformations.
GOC & STEREOCHEMISTRY
CSIR June 2011 Question and Answer
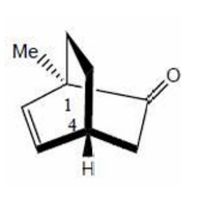
Explanation:
The substituents at each chiral centre are numbered according to the CIP priority rules. The atom with the highest atomic number is assigned the highest priority. After orienting the lowest priority group away from the observer, the sequence of remaining substituents is traced. Both stereocentres show a clockwise arrangement, leading to R-configuration at positions 1 and 4.
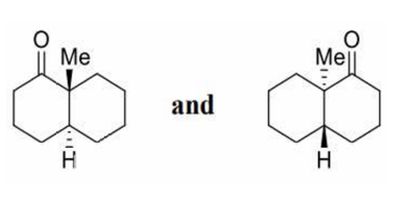
Explanation:
A 180° rotation about the carbonyl carbon–carbon bond converts one structure into the other. Since the two structures are interconvertible by simple rotation and no bond breaking is required, they represent the same molecule and are therefore identical, not enantiomers or diastereomers.
Explanation:
The substituents at each chiral centre are numbered according to the CIP priority rules. The atom with the highest atomic number is assigned the highest priority. After orienting the lowest priority group away from the observer, the sequence of remaining substituents is traced. Both stereocentres show a clockwise arrangement, leading to R-configuration at positions 1 and 4.
Explanation:
The t-butyl group is extremely bulky and therefore strongly prefers the equatorial position to avoid 1,3-diaxial interactions. Hence, at C-1 the t-butyl group is equatorial.
Since the substituents are trans-1,3-disubstituted, when one substituent is equatorial, the other must be axial. Therefore, the methyl group at C-3 occupies the axial position.
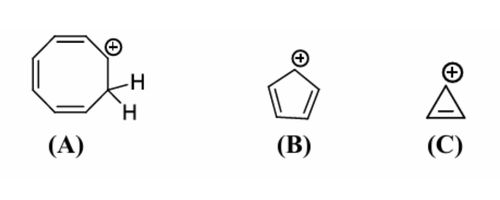
Explanation:
(A) shows homoaromaticity, where cyclic delocalization is maintained through-space despite interruption in conjugation.
(B) is a planar cyclic system with 4π electrons, which follows the 4n rule and is therefore antiaromatic.
(C) is the cyclopropenyl cation containing 2π electrons, satisfying the Hückel (4n+2) rule, making it aromatic.


