ABC Chemistry – Best Chemistry Coaching in Dwarka, Delhi
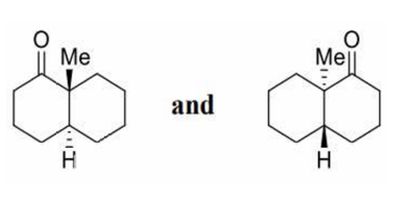
Explanation:
A 180° rotation about the carbonyl carbon–carbon bond converts one structure into the other. Since the two structures are interconvertible by simple rotation and no bond breaking is required, they represent the same molecule and are therefore identical, not enantiomers or diastereomers.
Explanation:
The substituents at each chiral centre are numbered according to the CIP priority rules. The atom with the highest atomic number is assigned the highest priority. After orienting the lowest priority group away from the observer, the sequence of remaining substituents is traced. Both stereocentres show a clockwise arrangement, leading to R-configuration at positions 1 and 4.
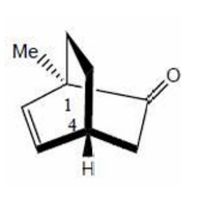
Explanation:
The substituents at each chiral centre are numbered according to the CIP priority rules. The atom with the highest atomic number is assigned the highest priority. After orienting the lowest priority group away from the observer, the sequence of remaining substituents is traced. Both stereocentres show a clockwise arrangement, leading to R-configuration at positions 1 and 4.
Years
Chemical Science Exams
Difficulty
Question Type
Subjects
Explanation:
The t-butyl group is extremely bulky and therefore strongly prefers the equatorial position to avoid 1,3-diaxial interactions. Hence, at C-1 the t-butyl group is equatorial.
Since the substituents are trans-1,3-disubstituted, when one substituent is equatorial, the other must be axial. Therefore, the methyl group at C-3 occupies the axial position.
(1) Cr(II)/Ru(III)
(2) Cr(II)/Co(III)
The rate of acceleration in going from an outer-sphere to an inner-sphere mechanism is lower for (1) relative to (2). Its correct explanation is:
CLASSES
Study Materials
Official Website Directory
Essential Link
User Policies & Support


ABC Chemistry
ABC Chemistry is the Best Chemical Science Coaching Centre for IIT-JAM, CSIR-NET, GATE and CUET PG, BARC, TIFR & UPSC Geo-Scientist exams. We provide high-quality classroom & online programs with expert faculty support.
Our structured learning, advanced study materials, regular tests & doubt-clearing sessions help students crack India’s top national level chemistry exams.
ABC Chemistryis a dedicated learning platform committed to building strong conceptual foundations in Chemistry for school students, competitive exam aspirants, and higher-education learners. Founded with a vision to deliver clarity-driven, result-oriented education, ABC Chemistry focuses on conceptual depth, exam relevance, and personalized mentorship.
At ABC Chemistry, we believe Chemistry is not about memorization—it is about understanding concepts, visualizing reactions, and applying logic. Our teaching methodology encourages questions, discussion, and analytical thinking, ensuring students gain long-term mastery rather than short-term marks.
To become a trusted name in Chemistry education by nurturing confident, conceptually strong students who can excel in academics, competitive exams, and research-oriented careers.
